
Pfizer's Transformative Science and Cutting-Edge Technology Are Protecting People of All Ages From Life-Threatening Infections and Cancer
At Pfizer, we discover and develop novel vaccines that help protect people around the world from deadly viruses, bacteria or cancer. In 2018, we focused on rational vaccine design — understanding the structure of proteins enabled by high-tech tools like cryo-electron microscopy as a basis to design and produce those proteins for use in vaccines.
A reflection of the importance of our work is the fact that two of our vaccines were awarded Breakthrough Therapy designations by the U.S. Food and Drug Administration (FDA): our 20vPnC vaccine candidate which entered Phase 3 development in December 2018 to help prevent invasive pneumococcal disease and pneumonia caused by 20 Streptoccocus pneumoniae serotypes in adults aged 18 years and older, and our Meningococcal Group B Vaccine which received licensure in 2014 to help protect against the potential life-altering consequences of invasive disease caused by Neisseria meningitidis serogroup B in adolescents and young adults, and is now being evaluated in children 1-9 years of age.
We are also advancing a late stage vaccine candidate that targets disease-causing toxins produced by Clostridium difficile, which has been recognized by the World Health Organization (WHO) as a significant medical problem and was designated as a priority pathogen by the U.S. Centers for Disease Control and Prevention (CDC). C. difficile is becoming a growing concern worldwide as cases have been increasingly reported beyond hospitals doors in the general community. C. difficile continues to be the most common cause of antibiotic-associated diarrhea in the health care setting, making it an important focus for prevention. Our vaccine candidate may help prevent disease in older adults who are at higher risk of infection, and if successful, could further address urgent antimicrobial resistance threats worldwide.
We also began the clinical trial to investigate an experimental vaccine to protect against respiratory syncytial virus (RSV), a virus that kills approximately 120,000 infants a year and is responsible for a substantial disease burden in adults. Our first cancer vaccine candidate continues to progress as well - we currently have a vaccine-based immunotherapy regimen (VBIR) in phase 1 that is being investigated in prostate cancer.
Vaccine R&D can be challenging. In 2018 we discontinued enrollment due to lack of efficacy in our Phase 2b trial, STRIVE (STaphylococcus aureus SuRgical Inpatient Vaccine Efficacy), which was evaluating the company's investigational Staphylococcus aureus (S. aureus) multi-antigen vaccine. Pfizer is evaluating next steps for the potential development of a S. aureus vaccine. While this development was disappointing, we believe that learnings may be derived from STRIVE that could help us to understand the reasons for the failure and inform the development of future vaccines.
We are optimistic that the scientific progress we have achieved in our vaccines pipeline in 2018 will lead to licensed vaccines and have a vital and global impact in protecting people from the suffering and fatalities caused by vaccine preventable infectious diseases.

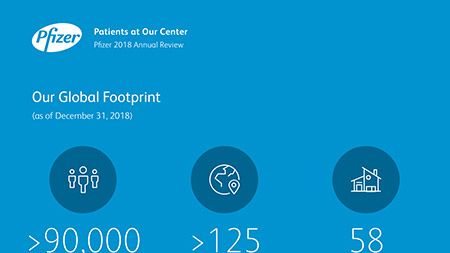

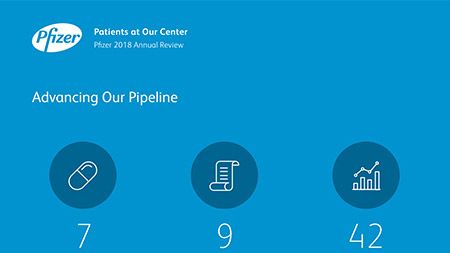



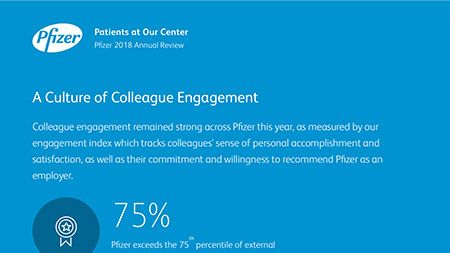
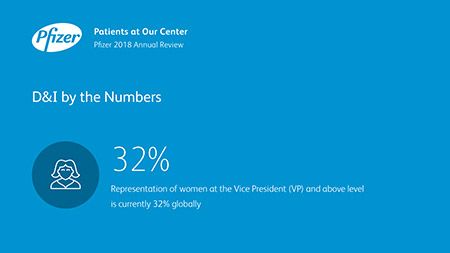




Ian Read: Contributions to Improving Human Health
Albert Bourla: Breakthroughs that Change Patients' Lives
Ushering in a New Era of Pfizer R&D Productivity
Advancing Our Leading JAK Science
Overcoming Therapy-Resistant Disease
Tackling Respiratory Syncytial Virus (RSV) Through Breakthrough Science and Technology
Catalyzing Innovations in Global Health
Contributing to the UN Sustainable Development Goals
Supporting Digital Health Start-Ups
Improving the Health of Women and Their Families