





Collaborating in new and dynamic ways with innovators across the health landscape is very important in our efforts to improve patients' lives. Our approach is creative yet disciplined, focusing on specific opportunities and the right partners to accelerate innovative science, improve operations and find better ways to deliver needed therapies. Increasingly, this has led to unique, focused alliances with our global pharmaceutical peers.
Over the last several years the health care landscape has seen tremendous scientific breakthroughs happening at Pfizer and beyond our walls. There are exciting opportunities to collaborate in new ways to augment our own discovery efforts. We look to bring together the best resources so that we can get the most impactful treatments and vaccines to patients, faster — it's what motivates us every day.
Our immuno-oncology alliance with Merck KGaA, Darmstadt, Germany continues to move forward quickly and demonstrate how effectively we are working together. Our co-development of avelumab, the fully human anti-PD-L1 IgG1 monoclonal antibody, continues to reach and surpass new milestones.
In 2015, the U.S. Food and Drug Administration granted Breakthrough Therapy, Orphan Drug, and Fast Track review designations for avelumab as a potential treatment for patients with metastatic Merkel cell carcinoma who have progressed after at least one previous chemotherapy regimen. These designations represent significant milestones in helping us bring this potentially important therapy to patients as quickly as possible. There is currently no therapy approved specifically for the treatment of metastatic Merkel cell carcinoma, a rare and aggressive type of skin cancer.
As part of the agreement with Merck KGaA, Darmstadt, Germany, the alliance is co-promoting Pfizer's anaplastic lymphoma kinase (ALK) inhibitor Xalkori® (crizotinib) in a number of markets including the U.S., Canada, Japan and five European Union countries (France, Germany, Italy, Spain and the U.K.). The agreement showcases our shared commitment to establishing a combined oncology sales organization in key markets in advance of the potential launch of avelumab-based treatment regimens in the future. Xalkori is the first ALK inhibitor approved in the U.S., Japan and the European Union and is supported by three positive global randomized trials in the first- and second-line ALK-positive metastatic non-small cell lung cancer treatment settings.
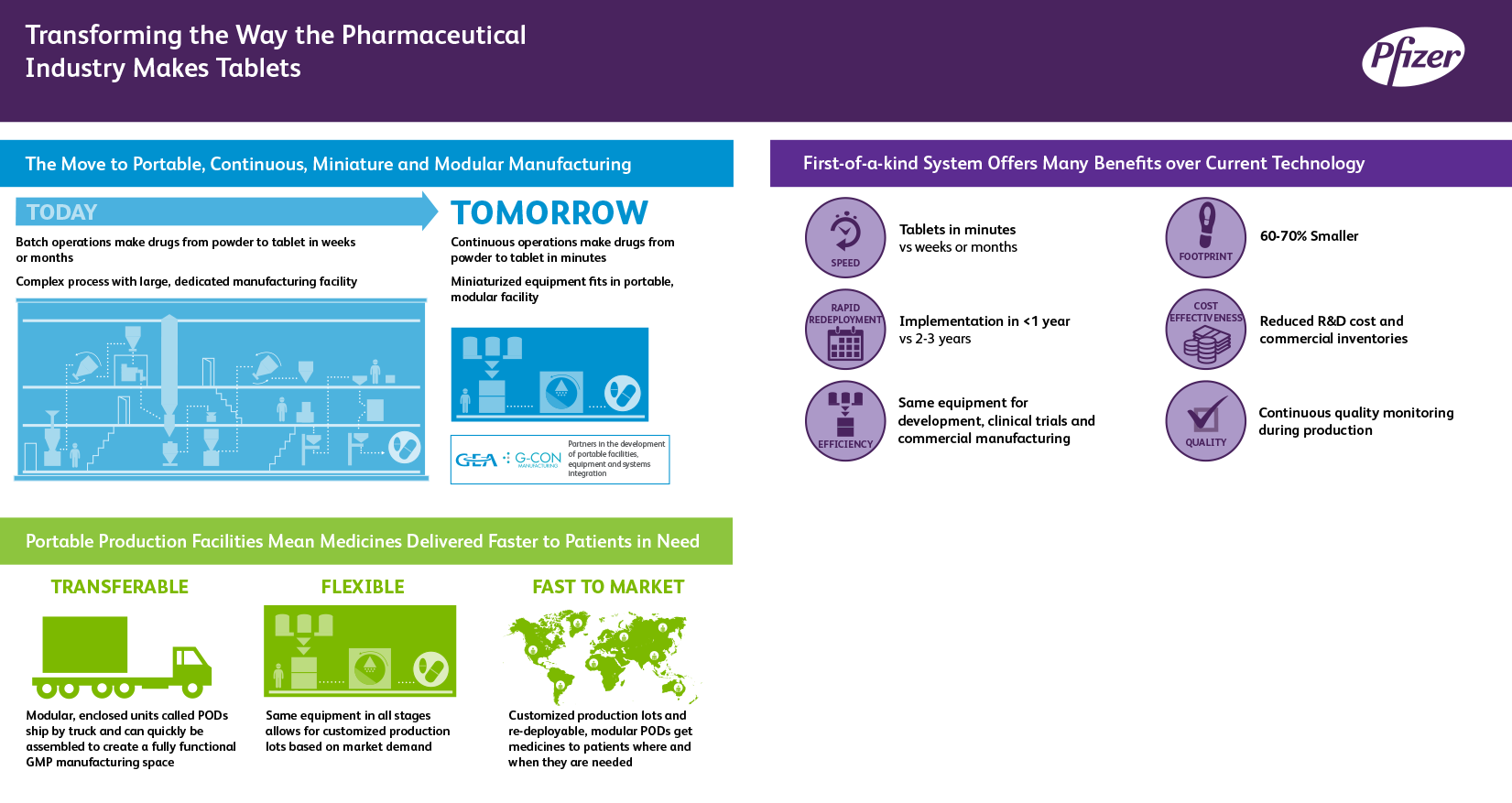
We have entered into a multi-year collaboration with GlaxoSmithKline PLC (GSK) to develop a next-generation design of our portable, continuous, miniature and modular (PCMM) prototype for oral solid dose pharmaceutical development and manufacturing. Pfizer's current PCMM prototype is an autonomous pod that may be quickly shipped from location to location and readily brought online to create a fully functional module that is compliant with industry-standard good manufacturing practice guidelines.
Together with GEA Group, G-CON Manufacturing and GSK, which have notable technical and regulatory experience in continuous processing, we will conduct coordinated experiments to create the next-generation design of our current PCMM prototype. This collaboration expands upon Pfizer's existing collaboration with GEA and G-CON Manufacturing, which resulted in the design of the prototype unit currently at Pfizer's labs in Groton, Connecticut, U.S.
The pharmaceutical industry has been trending toward lower volume products, driven by an increased focus on precision medicine approaches to develop and commercialize new therapies. This creates a need for smaller, more flexible continuous processing technologies.
PCMM has the potential to transform the current biopharmaceutical industry standard of using batch processing to manufacture tablets and capsules from powders — an oftentimes complex process that requires large, dedicated manufacturing facilities. The PCMM continuous process takes only minutes from the addition of raw materials to the completion of finished tablets or capsules.

We believe coupling Pfizer's industry-leading development and manufacturing capabilities with GSK's experience and expertise in continuous processing has the potential to lead to a superior technology, thereby allowing us to more quickly and efficiently bring therapies to patients.
Transforming the Way the Pharmaceutical Industry Makes Tablets
Pfizer and Eli Lilly and Company have resumed the Phase 3 clinical program for tanezumab, following a decision by the U.S. Food and Drug Administration to lift the partial clinical hold that had been in place for tanezumab and all other anti-nerve growth factor antibodies since 2012 due to adverse changes in the sympathetic nervous system of mature animals. (Studies in terminal cancer pain were allowed to proceed.) The decision followed a review of a robust body of nonclinical and clinical data characterizing the sympathetic nervous system response to tanezumab.
Tanezumab is a humanized monoclonal antibody that selectively targets and binds to nerve growth factor, a regulator of pain processing and sensitivity, thereby inhibiting this protein from activating pain-signaling neurons. Prior clinical studies of more than 11,000 patients compared tanezumab to placebo and other select commonly used pain medicines.
We're pleased to work with Eli Lilly to advance the Phase 3 program for tanezumab. If approved, tanezumab may offer an innovative, non-narcotic treatment for patients with certain pain conditions.
Sharing data to speed cancer research, Pfizer is helping to usher in a new era of data transparency. We have joined with industry and
research partners to challenge traditions and collaborate in new ways to help cancer patients get the greatest possible benefit from our
vast collection of clinical trial data. Project Data Sphere is an independent not-for-profit data-sharing initiative led by the CEO
Roundtable on Cancer Life Sciences Consortium. The overarching goal is to foster collaboration across companies and academia to advance
research that has the potential to improve outcomes for cancer patients. Pfizer has contributed data from studies in breast, lung, prostate
and colon cancer that had already been analyzed and used. Doing so gives the data a second life,
allowing other researchers to use them
in different ways that may lead to important potential new insights and discoveries. Researchers around the world will have access to these platforms,
with the hope that they will then generate new research hypotheses and accelerate innovation.
Aggregating these data sets empowers cancer researchers to potentially formulate novel research hypotheses and interrogate data in new ways.
With the rise of targeted therapies, a practical method for matching cancer patients with specific drug candidates is needed to enable the evolution of precision medicine. As such, Pfizer has entered into a long-term collaboration with Thermo Fisher Scientific and Novartis to develop a multi-marker, universal next-generation sequencing (NGS) oncology test panel that will serve as a companion diagnostic (CDx) initially for non-small cell lung cancer (NSCLC) followed by other cancer indications across multiple development programs. NGS enables testing of multiple genes simultaneously from a single tumor sample to help to identify their unique genetic profile. The ultimate goal is to use this information to guide the appropriate therapy choice among multiple drug candidates. NGS also has the potential to improve safety, effectiveness and health outcome of patients via targeted risk stratification and tailored treatment approaches. The collaboration, focused on a universal testing approach, could also accelerate the development and registration of several new NSCLC drugs and other drug indications, with the ultimate goal of providing patients greater access to more targeted treatments and appropriate clinical trials as quickly as possible. It is anticipated that the NGS test panel being developed may have the potential to receive simultaneous approval for several genes from the U.S. Food and Drug Administration and will be used as a CDx for multiple drugs.
With scientific progress moving at an ever-accelerating pace, how can we ensure that the patient perspective and patient needs stay front and center? To find out, Pfizer hosted the Global Oncology Advocacy Innovators Meeting (GO AIM), a first-of-its kind event that brought together cancer patient advocacy leaders from around the globe. Patient advocates shared with Pfizer that they are looking for a true partner. From helping to design clinical trials to educating policymakers on the value of potential new cancer innovations, patients and their advocates can provide important perspectives and direction at even the earliest stages of research and development.
We are at a point where research is flourishing, and we are bringing new thinking and hope to cancer patients. However, we must not stop there. We must continue to innovate. Partnering with patient organizations is critical to further explore the needs of cancer patients around the world and together find potential solutions to address the challenges they face.
Pfizer has signed on to the Dementia Discovery Fund, a new initiative that aims to boost investment in developing novel treatments for dementia. The fund is managed by SVLS Venture Partners and brings together Alzheimer's Research UK, the U.K. government and six pharmaceutical companies (Pfizer, Biogen, GSK, Eli Lilly, Johnson & Johnson and Takeda) with the goal of financing early stage drug development projects. More than $100 million has already been raised to develop pioneering new medicines.
Neuroscience research has been a particularly challenging area for significant advances, due to the brain's complexity. The science required to deeply understand its function is daunting and difficult. Yet we are seeing important scientific momentum in advances that are helping us unravel the underlying molecular disease pathophysiology of dementia and other neurological conditions. Cross-sector collaboration is a critical success factor to deliver unique and transformative potential therapies for patients.
Because of the significant social, financial and scientific challenges that dementia-related illnesses pose, we believe that it will be most beneficial for patients if we create, identify and support innovative ways to partner with other pharmaceutical companies to help tackle dementia.
