Our business, our purpose
Global Businesses – PEH
Pfizer Essential Health (PEH) medicines reach more than 300 million patients a year at virtually every stage of life. The depth and breadth of our portfolio of trusted medicines and capabilities delivers value to patients throughout the world. Whether it’s using new technology and our deep experience to increase access to an essential medicine, or combating an emerging health risk, our ability to innovate and anticipate customer and patient needs allows us to make meaningful and inspired contributions to improving global health.
PEH Market Leadership: Global Reach and Impact
Global leader in anti-infectives
Number one in total biosimilar sales
Leading company in global off-patent sterile injectables
Recognized leader in emerging markets
Changing the Face of Patient Care Through Biosimilars
As one of the world's leading innovative biopharmaceutical companies, Pfizer is at the forefront of developing, manufacturing, and delivering high-quality biosimilars to patients, physicians and payers. Biosimilars have the potential to expand patient access to treatment and to contribute to more sustainable health care systems. This should be achieved without compromising quality and by making resources available for further investment and innovation so that more patients may receive the best possible care.
At Pfizer, we are making high-quality versions of biologic medicines available to patients and physicians in a number of countries around the world across multiple life-threatening and chronic diseases in the fields of inflammation, immunology, and oncology. Our work in biosimilars, which reflects both our commitment to scientific innovation and our focus on patient access, is a prime example of how we are driving value in every aspect of our business to improve patient health.
We have 10 years of global experience in biosimilars. Our current commercialized in-market portfolio includes Inflectra® (infliximab-dyyb in the U.S.), the first biosimilar monoclonal antibody to be approved in Europe and the U.S., and Nivestim® (filgrastim) and Retacrit® (epoetin zeta), both approved in Europe. In December, the U.S. Food and Drug Administration (FDA) approved Ixifi™ (infliximab-qbtx), our second biosimilar to Remicade® (infliximab). Our pipeline is one of the largest globally, with 12 assets in various stages of development, including seven assets in mid- to late-stage development, and a number of compounds in registration in Europe, the U.S., and other areas of the world. These include potential biosimilars for some of the world’s most well-known biologics in the treatment of cancer, including Herceptin® (trastuzumab) and Avastin® (bevacizumab).
Read more about how our biosimilars are transforming patient access to care
Addressing Global Public Health Needs with Anti-Infectives
At Pfizer, we are driven by our desire to protect global public health and reduce the suffering caused by infectious diseases. Starting with our pioneering work on penicillin in the 1940s, we have a long and proud heritage of addressing evolving infectious disease challenges. We offer the industry’s largest and most diverse portfolios of anti-infective therapies, which includes more than 80 potentially life-saving medicines.
We continue to invest in novel treatments for infectious diseases to address the greatest patient medical needs and provide solutions that go beyond the medicine. Working together with the infectious disease community, we are striving to overcome the challenges associated with antimicrobial resistance – one of the largest threats to global public health today – by leveraging our expertise and capabilities to share solutions with our healthcare partners.
Read more about how Pfizer is innovating to deliver new anti-infective therapies
Ensuring Access to High-Quality Sterile Injectables
Pfizer has one of the broadest and most diverse portfolios of important, difficult-to-manufacture and life-saving sterile injectable medicines in the industry. Through this portfolio of more than 250 products, we are firmly positioned to support many areas of hospital care and make a deep and meaningful impact on patients, now and in the future.
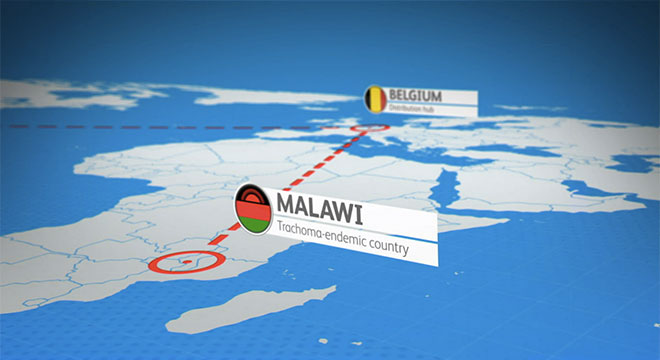
Leveraging Science to Create Supply Chain Solutions
Pfizer's goal is for patients to receive uninterrupted access to our medicines.
We continue to use our operational and supply chain data from enterprise systems to identify processes to better meet the needs of our patients. In 2017, this information helped us address drug shortages of several injectable medicines used in hospitals and other clinical settings. Within Pfizer Global Supply, our manufacturing teams work with health care bodies to put solutions in place to help expedite supply recovery for important medicines. One critical element of Pfizer supply recovery activities has been providing scientific data to regulatory authorities to support extended-use dating for products in the supply chain. We are also working to minimize supply shortages in the future by investing in advanced technology and capabilities.
Bringing Pfizer's Trusted Legacy Medicines to Patients
Global brands are medicines that treat a range of conditions and have been part of a legacy that has made Pfizer one of the most trusted names in medicine. Although many of these medicines are no longer exclusively manufactured by Pfizer, they often remain important treatments for patients in the developed world and are beginning to be part of core treatment regimens in emerging markets. Rigorous studies over decades have established the value that these medicines bring to the health care community. We also provide a range of generic therapeutic options to patients and health care providers and payers.
Overall, our portfolio of medicines includes treatments for:
- Cancer
- Cardiovascular disease (high cholesterol, high blood pressure)
- Central nervous system disorders (depression, anxiety)
- Deep venous thromboembolism
- Erectile dysfunction
- Infectious disease
- Inflammation
- Pain (neuropathic, fibromyalgia, osteoarthritis)
- Women’s health (menopause, family planning)
A New Chapter for an Iconic Brand
Since its first approval in 1994, our iconic erectile dysfunction treatment Viagra® (sildenafil citrate) has been one of our all-time best-selling medicines. Importantly, Viagra revolutionized the way we think and talk about sexual health. On December 11, 2017, the patent exclusivity of Viagra expired in the U.S. Pfizer Essential Health will continue to offer branded Viagra, and Greenstone, a wholly-owned subsidiary of Pfizer with a comprehensive and diverse product portfolio of generic medicines, launched an authorized generic sildenafil citrate. Both products will come with the assurance of our quality manufacturing standards. In addition, in November, following an extensive review process, the U.K. Medicines and Healthcare products Regulatory Agency (MHRA), approved the reclassification of sildenafil citrate 50mg as a non-prescription pharmacy medicine. Pfizer expects to introduce the pharmacy-supplied product, Viagra Connect® in the U.K. in spring 2018.
How our work in this area is supporting the Sustainable Development Goals
Goal : Good Health and Well-Being
Explore our passion in action