Our Innovation
Vaccines
Pfizer Vaccines is infusing the power of transformative science into disease prevention. Our colleagues are committed to pursuing research and forming partnerships that speed the development and delivery of new, life-saving prophylactic vaccines for bacterial and viral diseases, infectious diseases and even cancer.
Taking on the Challenge of Pneumococcal Disease
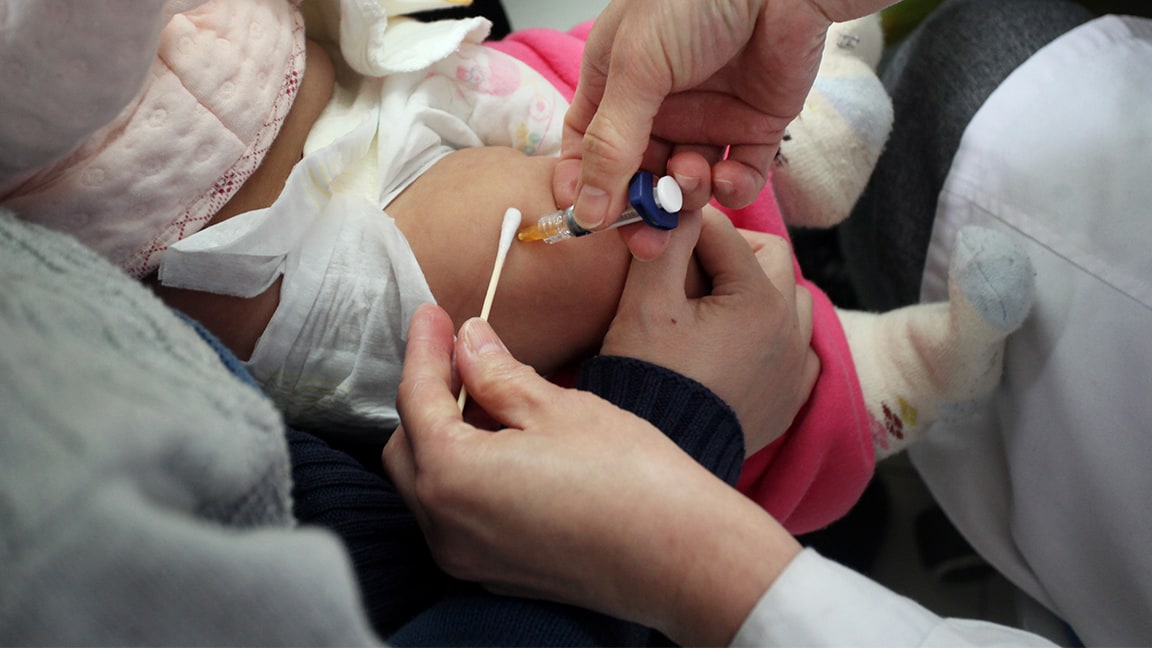
In 2017, Prevenar 13® (Pneumococcal 13-valent Conjugate Vaccine [Diphtheria CRM197 Protein]) was introduced to India’s Universal Immunization Program (UIP), with support from Gavi, the Vaccine Alliance, to help reduce the number of pneumonia deaths in children five years of age and younger in India. An estimated 105,000 children in India 5 years of age and younger died in 2010 because of pneumococcal pneumonia, the highest number in the world.1
To date we delivered more than 300 million doses of Prevenar 13, helping to protect 20 million babies in Gavi countries each year. Pfizer also introduced a multi dose-vial presentation of Prevenar 13 to Gavi countries in 2017.

We believe the introduction of Prevenar 13 over a three-year period should have a significant impact in lowering the burden of pneumococcal disease and potentially help protect more than five million infants across the five Gavi-funded states of Himachal Pradesh, Bihar, Uttar Pradesh, Rajasthan and Madhya Pradesh, which represent about 20 percent of India’s birth cohort. With this, India joins more than 100 countries to have included Prevenar 13 in their pediatric National Immunization Program.
Read more about our work with Gavi
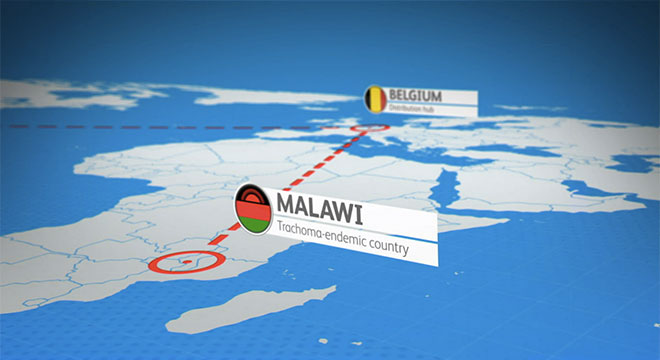
Pfizer is dedicated to meeting the needs of people impacted by humanitarian emergencies. In 2017, through our expanded humanitarian assistance program, hundreds of thousands of doses of Prevenar 13® were distributed in Africa and the Middle East to vaccinate those in unfortunate situations qualifying as humanitarian emergencies under World Health Organization (WHO) criteria.
Pfizer also provides support for programs and disease awareness. In Niger, Mali and the Democratic Republic of Congo, Pfizer supports health care worker training intended to help people recognize the early signs of pneumonia and inform caregivers of the importance of vaccination. Plans are in place to extend this training to Angola.
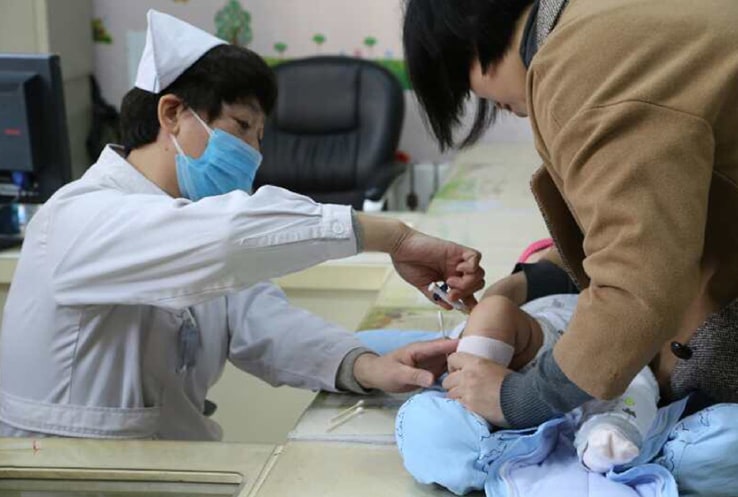
Pfizer also launched Prevenar 13® in China in 2017 to help protect children 6 weeks to 15 months of age, with the vaccine now available in 22 provinces. Approximately 30,000 children in this age group in China die due to pneumococcal diseases every year.2
Combating Meningitis in Infants, Adolescents and Young Adults
Meningococcal disease continues to be a global health burden – and Pfizer remains dedicated to helping to protect against this disease. Changing epidemiology continues to spur outbreaks of this disease, and we continue to help supply vaccine to the areas in greatest need. In 2017, we responded to emergencies by supplying vaccine to outbreaks, including those in Nigeria, Australia and parts of Europe.
We continued to advance Nimenrix® (meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine) with expanded indications in many parts of the globe to include infants. As we continue to build out Nimenrix manufacturing capabilities we have invested in our manufacturing sites, including Puurs, Belgium, Grange Castle, Dublin, Ireland and North Carolina, U.S.
Read more about manufacturing sites and how we are bringing jobs to North Carolina.
In May, the European Commission approved Trumenba® (Meningococcal Group B Vaccine) for the prevention of invasive meningococcal disease caused by Neisseria meningitidis serogroup B (MenB), in both a two- and three-dose series, in individuals 10 years of age and older, with subsequent approvals in Australia and Canada (in Canada, for individuals 10 through 25 years of age). Adolescents and young adults are a critical demographic for vaccination against MenB due to inherent environmental and social risk factors such as close-quartered living and sharing behaviors. While MenB is uncommon, the disease can progress rapidly, and symptoms are difficult to distinguish from other more common infections, with flu-like symptoms such as headache, nausea and vomiting among the earliest signs.
We also made significant progress in expanding the reach of this important vaccine, with Trumenba launches in seven countries, including the U.K., Malta, Germany, Denmark, Norway, Finland and Portugal. The broader availability of Trumenba reinforces our dedication to advancing important vaccines that can help protect adolescents and young adults, a population at increased risk for meningococcal disease.
Progressing Our Pipeline to Take on the Global Public Health Burden of Infections
Clostridium difficile
Clostridium difficile (C. difficile), is the most common cause of antibiotic-associated diarrhea in the health care setting and an increasing concern worldwide. The bacteria are present in the environment and can colonize the human intestine. When conditions are right, such as when patients have taken antibiotics, they can multiply and express toxins that lead to diarrhea. A recent study from the U.S. Centers for Disease Control and Prevention (CDC) found that the incidence of C. difficile infection (CDI) nearly doubled from 2001 to 2012, and in 2011 alone there were 453,000 cases in the U.S. and approximately 29,000 deaths. In light of the burden of disease, the CDC classified C. difficile as an urgent public health threat in 2013.
Two main toxins cause this illness, and we are progressing PF-06425090, our vaccine candidate comprised of detoxified versions of these toxins in clinical trials. We used a novel genetic engineering approach to make the vaccine. In 2017, we initiated the Phase 3 CLOVER (CLOstridium difficile Vaccine Efficacy tRial) trial to assess the vaccine candidate’s safety and efficacy in preventing infection in adults.
Staphylococcus aureus
Likewise, we continue to investigate our Staphylococcus aureus (S. aureus) multi-antigen vaccine [SA4Ag] candidate. S. aureus is a bacterium that approximately 30 percent of healthy individuals may be colonized with at any given time. In health care settings, these infections can be serious or even fatal. Our vaccine is designed to help protect against invasive S. aureus infections by inducing functional antibodies that intercept and help to kill the bacteria when penetrating our natural barriers. The Phase 2b STRIVE (STaphylococcus aureus suRgical Inpatient Vaccine Efficacy) safety and efficacy study, investigating the potential of SA4Ag to help prevent invasive S. aureus surgical site infections occurring 90 days after surgery, is ongoing. The study is being conducted in 6,000 adult patients undergoing elective instrumented spinal fusion surgery.
Group B Streptococcus
In June, we initiated a Phase 1/2 trial of PF-06760805, our conjugate vaccine candidate to help protect against Group B Streptococcus (GBS) infection, which can manifest as a serious neonatal blood infection (sepsis), pneumonia and meningitis in newborns, with potentially fatal outcomes or long-lasting neurological damage in those infected. Women who are colonized with the GBS bacteria may pass it on to their newborns during labor and birth. This is the first clinical step in our efforts to explore the potential of immunizing women during pregnancy, a process known as maternal immunization, to protect their new babies from devastating infections.
Embracing Technology to Educate on Vaccination
Social media continues to transform the way people access health information, and Pfizer has eagerly embraced this technology to engage and educate people on the importance of disease prevention and the value of vaccination. As part of our patient-centric approach, we tap into social and digital channels to reach our key audiences. Whether it’s reaching adults via Facebook Live streaming videos focused on the importance of vaccination or educating though impactful videos, we strive to find new and innovative ways to help inform our audiences.

Pfizer’s Susan Silbermann, Global President and General Manager, Pfizer Vaccines, partnered with the United Nations (UN) Foundation’s Shot@Life at the UN General Assembly to highlight the impact of vaccination for children in developing countries.
Legendary entertainer Patti LaBelle joined Freda Lewis-Hall, M.D., Executive Vice President and Chief Medical Officer, Pfizer, in support of our Get Old initiative to encourage people to live well and stay in control of their health. Patti also encouraged adults 65+ to get a "New Attitude" about the risks of developing pneumococcal pneumonia and the importance of vaccination. Watch to learn more.
The Hero project also highlighted the value of staying healthy as you get older. It centers on the unique relationship between grandparents and grandchildren in Europe and the importance of protecting your heroes. In France, Precious Moments demonstrates the importance of vaccination to new parents through an engaging and relatable narrative.
Reference
1. Farooqui H, Jit M, Heymann DL, Zodpey S (2015). Burden of Severe Pneumonia, Pneumococcal Pneumonia and Pneumonia Deaths in Indian States: Modelling Based Estimates. PLoS ONE10(6): e0129191.
2. O'Brien KL, et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet. 2009; 374(9693): 893-902.
Explore our science in action