From Math to Medicine: What Are Mathematical Models and How Do They Predict Pharmacology?
It sounds strange, but predicting how medications impact diseases and the body is a lot like forecasting the weather.
To understand why, imagine opening a weather app on your smartphone to check the forecast for the upcoming weekend. You see that it’ll be sunny, 72 degrees, with a slight breeze—perfect weather. But how did scientists predict those precise physical conditions so far in the future?
The answer: mathematical models.
Scientists across disciplines use mathematical models to make predictions and understand complicated real-world systems, like the weather, traffic patterns, and biology.1 For meteorology, experts collect data about the weather from satellites, observation stations, and other measuring devices. Then they enter that information into equations that describe the factors that determine the weather, like temperature and wind speed. By solving these equations, meteorologists produce the forecasts that end up on our weather apps.2
In the same way, scientists build models to study human diseases and potential treatments beforea single patient takes a new medicine. This is called quantitative systems pharmacology (QSP), a field in which mathematical models simulate complex biological and disease processes.
Quantitative system pharmacologists build mathematical models with equations that describe the behavior of medicines, diseases, and the body, explains CJ Musante, VP of Scientific Research and QSP Head at Pfizer. They can then input new medications or dosages into the model and safely study how they might affect a patient, just like meteorologists forecast the weather.
But how does it all work? To better understand how experts turn mathematics into medicine, we spoke with Musante about the basic steps to create, test, and run a quantitative systems pharmacology model.
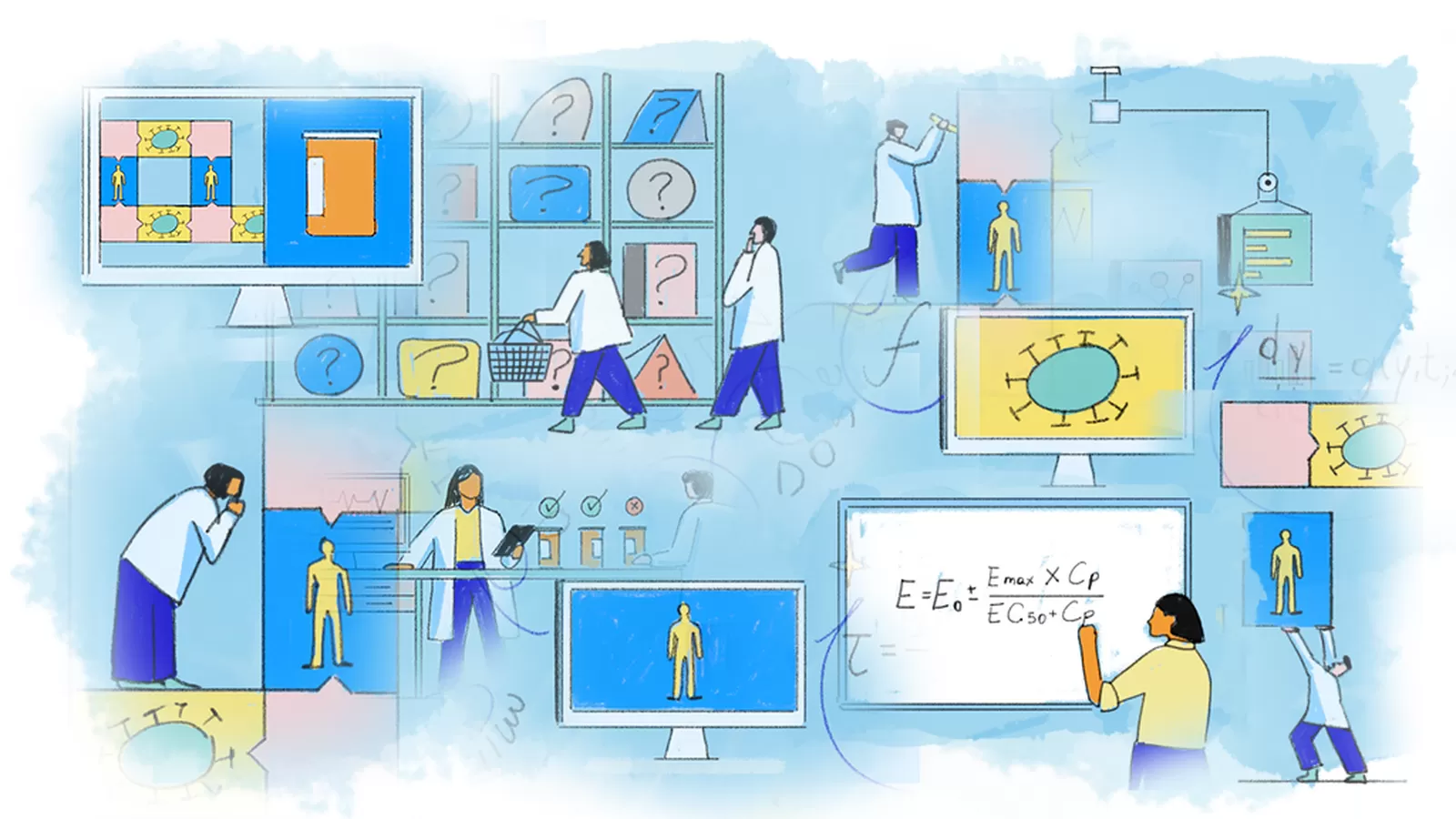
Step 1: Select the focus
Scientists choose what they want to study and define the question they want to answer. Then they drill deeper by selecting which medication, bodily processes, and health outcomes to focus on, sometimes using artificial intelligence to comb data sets and identify the best options.
Step 2: Set the rules
Scientists collect data and knowledge about the disease, medicine, and bodily processes. They often consult medical journals and human biology and physiology textbooks to unearth that valuable information. Quantitative systems pharmacologists also scour publications and databases to define how biological or physiological values may vary from person to person, like potential patients’ age, weight, or blood test results. This step establishes a solid foundation for the mathematical model to represent different patients and helps keep future predictions within the range of reasonable expectations.
Step 3: Craft equations
Next, experts write equations to describe the complex interplay between the medicine, disease, and body. These calculations might explain how a diabetes medication or the interaction between a cancer treatment and a tumor. Some equations help predict how quickly disease conditions might improve over time with treatment. Others describe the spatial attributes of the biological processes, such as how much a tumor may shrink in size. These computations are the QSP model’s building blocks.
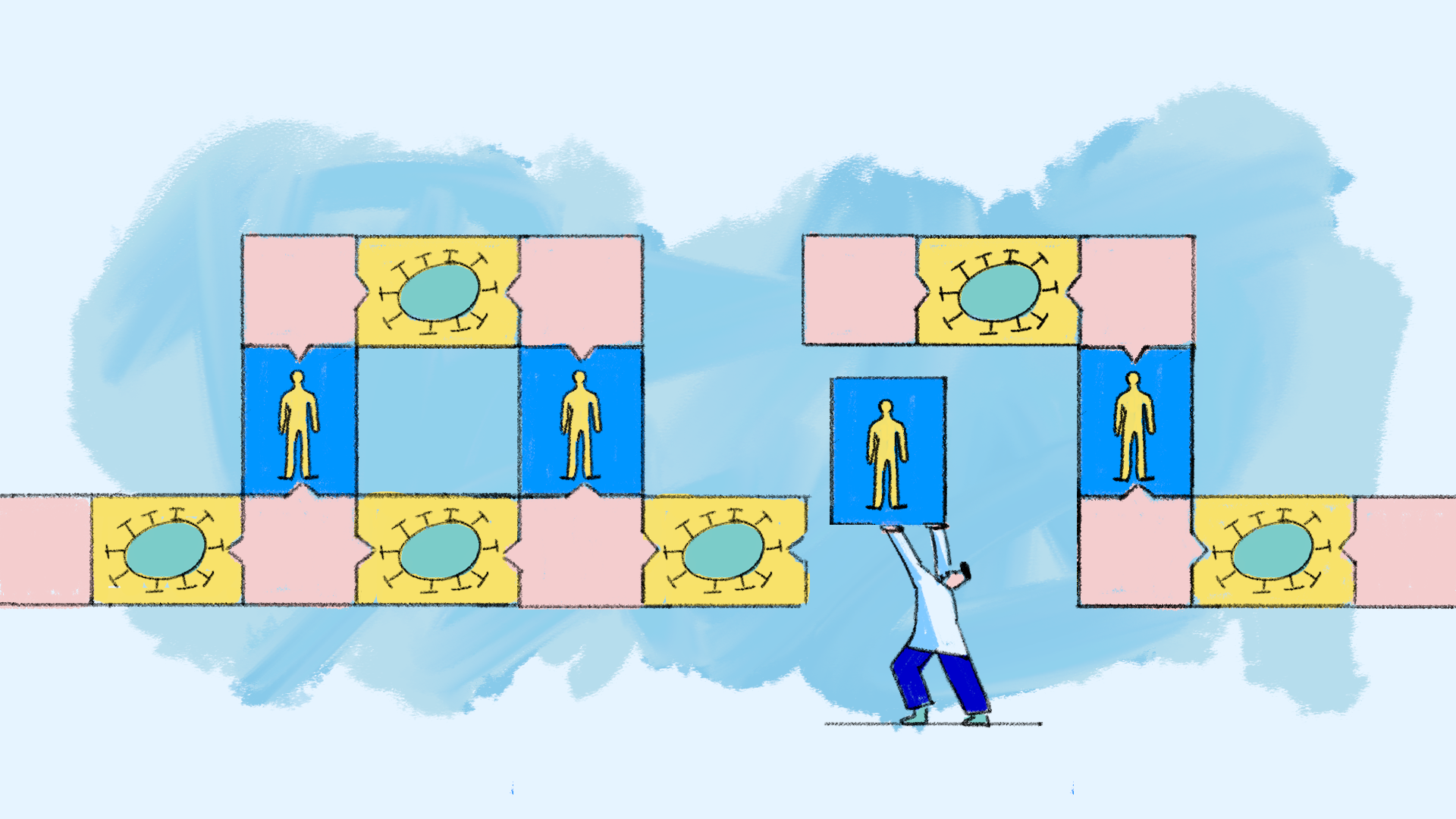
Step 4: Build a disease model
After that, scientistscombine the equations that describe the disease and its impact on the body into a single mathematical model. Often, this model helps reveal the characteristics or functions of a disease that scientists should target to produce the best health outcomes.
Step 5: Build a medication model
The scientists do the same for the equations describing the treatment and how it interacts with the disease and the body.
Step 6: Combine both models
Scientists merge both mathematical models and check the complete version to ensure it fits within the parameters defined in Step 2. Now, they have a working model and can begin honing its structure to deliver the most accurate forecasts.
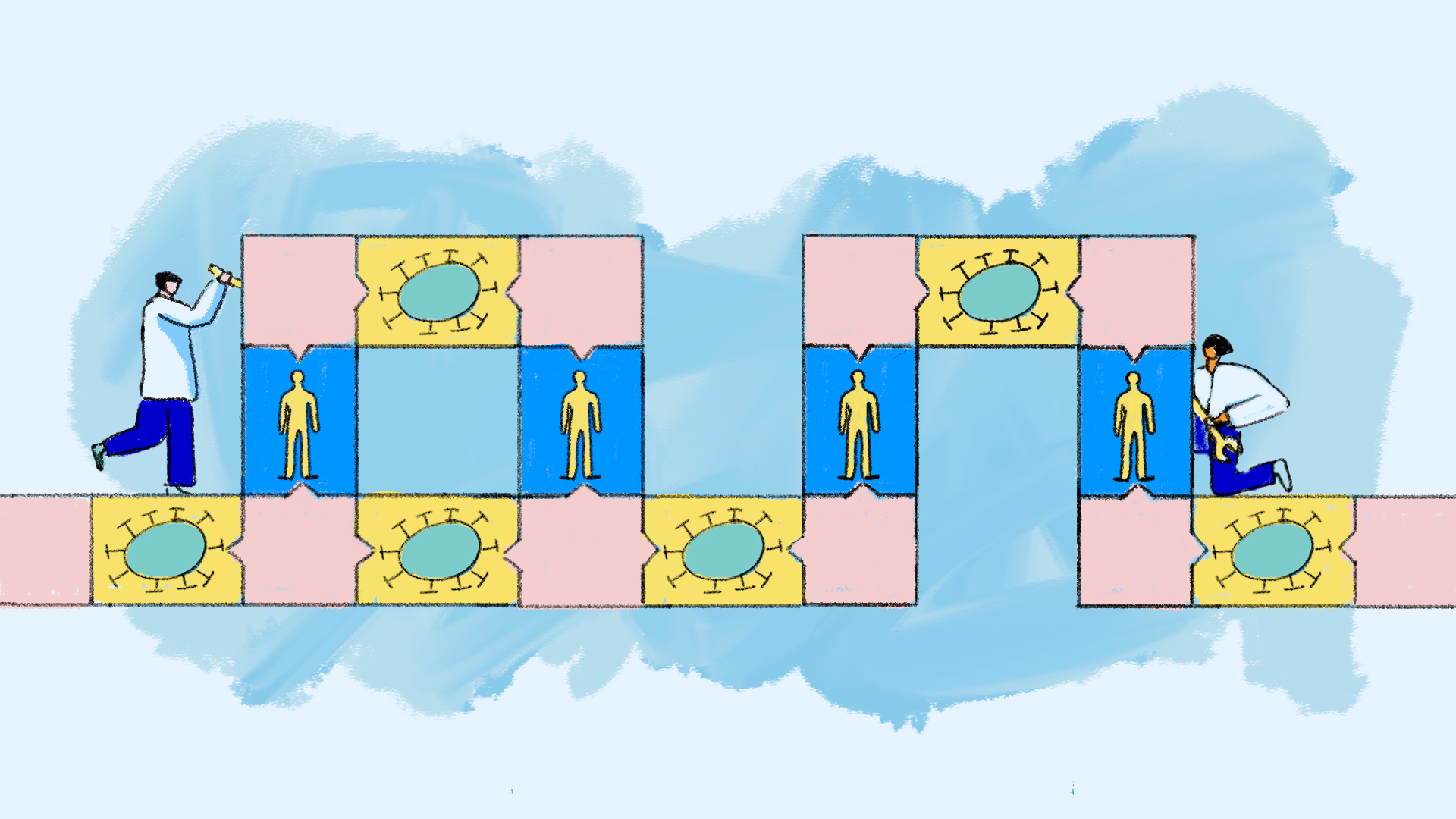
Step 7: Calibrate
Next, scientists fine-tune the model, often with data from past studies. Scientists frequently refer to this as the "training dataset." This helps them align the model’s projections with known real-world results. In special cases, scientists can adapt the model to suit the characteristics of specific patient groups to support precision medicine.
Step 8: Validate
Scientists then validate the model by running a final check with data that wasn’t used in the calibration process. Scientists refer to this as the "testing" dataset." This provides confidence that the model is capable of predicting new scenarios.
Step 9: Run studies
Scientists can now use the model to test entirely new medicines or doses. This allows scientists to quickly simulate a range of options before designing long-term clinical trials that require patient participation. Experts sometimes refer to simulated trials as “in silico,” meaning they occur solely within a computer, and involve simulated "virtual patients."
Step 10: Study the results
Scientists use the model’s results to support treatment development. Quantitative systems pharmacology has helped predict how well a new medicine will do its job, identify the right dose for medication, understand the characteristics of patients who may benefit most from treatment, and get life-changing medications to patients faster.
The future of forecasting
It took centuries for weather forecasting to develop from the theories of ancient Greek philosophers to the precise science it is today.3 Quantitative systems pharmacology didn’t suffer the same growing pains as it rapidly evolved into a critical branch of medical research. And if you had to forecast the discipline’s future, chances are it will come to play an even more significant role in supporting patients everywhere.
References
1. "Mathematical model." Encyclopaedia Britannica. https://www.britannica.com/science/mathematical-model. Last updated April 11, 2023. Accessed April 25, 2023.
2. "About Models." National Weather Service. https://www.weather.gov/about/models. Accessed April 25, 2023.
3. "History of weather forecasting." Encyclopaedia Britannica. https://www.britannica.com/science/weather-forecasting/History-of-weather-forecasting. Last updated Jan. 17, 2019. Accessed April 25, 2023.
![]()