How This Once Little-Known
Molecule Is Disrupting Medicine
By Sachi Fujimori | June 2022
With its proven success in vaccinations during the global pandemic, messenger ribonucleic acid, or mRNA, technology has hit prime time. The versatile platform is already being used to develop investigational vaccines against other infectious diseases. But its potential doesn’t end there. Over the next decade, this technology could transform medicine.
Many of the advantages of mRNA technology—its flexibility, speed, and potency—have scientists around the globe excited about its potential future applications, such as vaccinating against other pathogens, treating rare diseases and cancer, and more.

The unique thing about Pfizer is that we have experience with mRNA technology, but we also know the biology of disease, which is essential to harnessing the technology to make an impact on patients. No company on the planet is making mRNA at this scale and has done it with this speed.
Continuing its partnership with German biotechnology company BioNTech, Pfizer is developing potential mRNA vaccines against the seasonal flu and shingles, a painful illness related to chicken pox. Pfizer scientists are also working to apply mRNA technology to provide potential treatments for rare diseases.
Turning your body
into medicine factories
Scientists are looking to apply mRNA across a broad range of diseases. What makes it so versatile? The answer comes down to two basic biological principles:
Our cells are super protein factories.
They produce tens of thousands of different proteins that perform all the work in our bodies— from the enzymes that help us digest food to the fibers that make our muscles move.
Our cells are prolific protein factories.
And mRNA plays a key role in this process.
Messenger RNA is a molecule found in every living being. If DNA is our instruction manual for life, then mRNA is a single-page photocopy from this book—carrying genetic information for a specific protein.
Using these mRNA instructions, our cells make the proteins we need to sustain life.
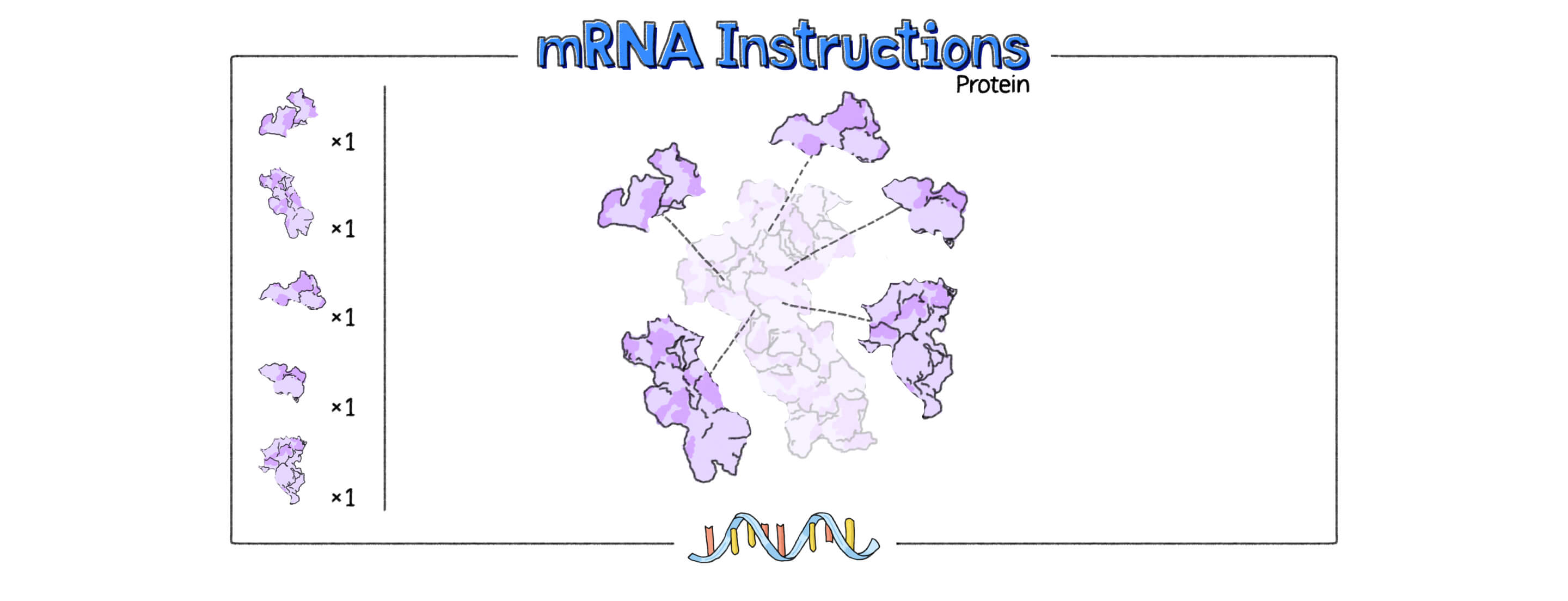
mRNA instructions can be coded with gene-editing proteins, tools that could repair single-gene mutations.
Proteins can also be medicine.
The core idea behind mRNA technology is to leverage the body’s natural protein-making abilities to make molecules that can help prevent or treat diseases. In this sense, mRNA turns our cells into factories that can produce proteins.
Depending on the target disease, mRNA instructions are tailored to encode a specific protein that may help prevent or treat a disease. These proteins, for example, may be useful in helping to fight cancer or teaching the immune system to recognize a specific virus.
Another key trait of mRNA is that it is very short-lived. Shortly after your cellular machinery uses it to make a protein, it’s destroyed. It doesn’t linger in the body. And because it doesn’t enter the cell’s nucleus, it can’t interact with your DNA.
“mRNA is like the instructions that come with a DIY furniture shipment. You basically use the instructions to build the furniture and once you’re done with the assembly, you can throw the instructions away,” says Pirada Suphaphiphat, Vice President of Viral Vaccines at Pfizer.
mRNA instructions are fleeting—they quickly self-destruct after being read.
Swift and nimble
In the early weeks of the pandemic, researchers were able to genetically sequence the SARS-CoV-2 virus, making it possible for Pfizer and BioNTech to hit the ground running in their quest to develop a vaccine against COVID-19.
“Once the virus is sequenced, you can quickly go and make the mRNA instructions in the lab. And that’s why it enabled a certain amount of speed that was not permissible with conventional vaccine approaches,” says Fernando.
Another benefit of the mRNA approach is that scientists don’t need to use the actual virus to make the vaccine. mRNA vaccines simply contain instructions, or genetic code, to make a copy of the novel coronavirus’s spike protein, which triggers an immune response in our bodies.
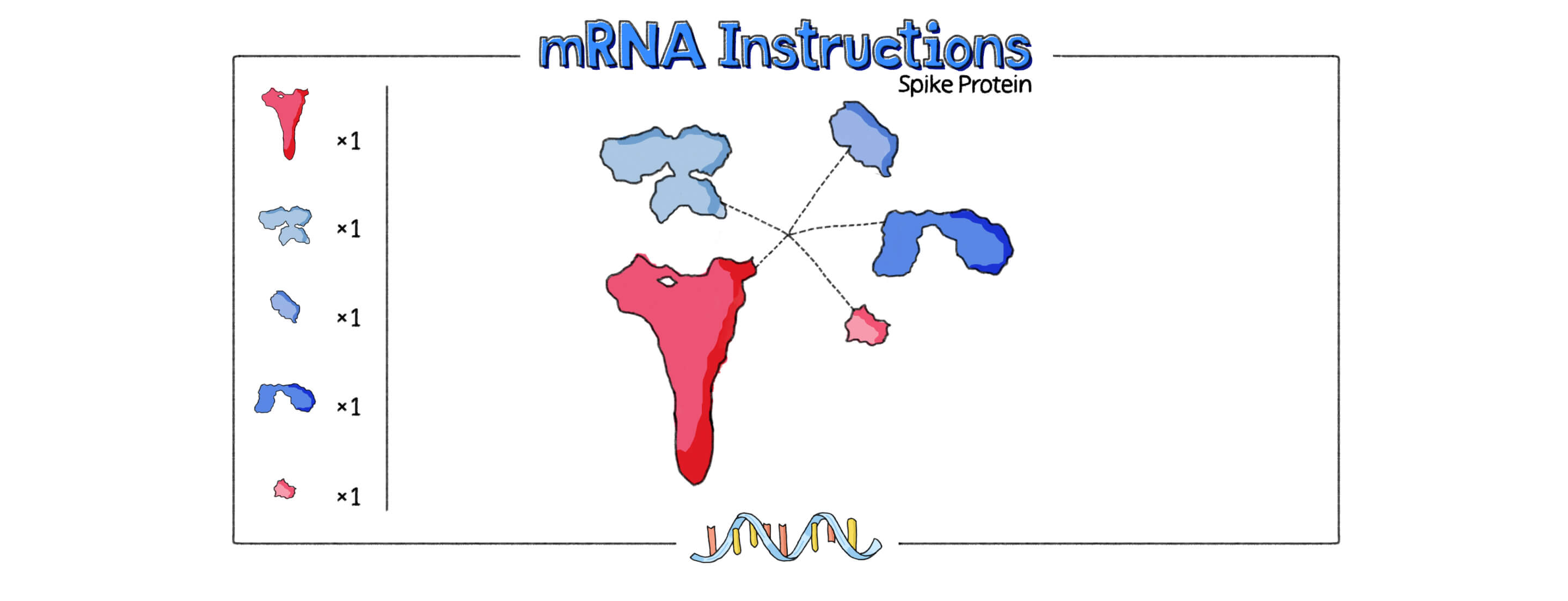
mRNA vaccines contain the instructions for making the SARS-CoV-2 spike protein.
The mRNA is packaged inside a lipid nanoparticle (LNP), a tiny bubble of fat, that serves as a delivery vehicle to get the molecule inside our cells.
When injected into the body, these mRNA instructions temporarily turn our cells into protein-making factories, which in the case of a vaccine, helps our body prevent disease.
Our cells use these instructions encoded by the mRNA included in the vaccine to make the SARS-CoV-2 spike protein.
Our cells produce the spike protein, which teaches our immune system to recognize and fight the virus.
Our cells display the spike protein like a “wanted poster”—teaching the immune system to recognize and protect against the virus.
As new strains of the novel coronavirus continue to emerge and we look ahead to future pandemic preparedness, the mRNA platform has another key advantage: It’s extremely nimble.
Scientists can obtain new instructions for making the virus's spike protein and “plug” them into the mRNA vaccine for testing and development.

The mRNA vaccine has a much more simplified manufacturing process, with a smaller footprint. This is a benefit that should help us respond to pandemics and to diseases that require regular strain updates like the seasonal flu.
Beyond the finish line
While mRNA is having its moment right now, it’s certainly not new. “Scientists have been working on mRNA medicines and vaccines for decades. It wasn’t born overnight,” says Fernando. “It’s just that these vaccines came to the finish line during a pandemic when the world really needed it.”
Pfizer scientists are eager to build upon this research foundation—and mRNA’s historic impact—to help even more patients.
What’s next for mRNA?
