How Finland’s Unique Genetic Heritage Is Being Used to Study the Links Between Genes and Diseases

The unique genetic heritage of the Finns — marked by repeated population bottlenecks and isolation from their neighbors in northern Europe — is helping scientists embark on a search for the complex links between genes and diseases.
Finland also has a robust network of biobanks, and the country has passed laws that make the voluminous biobank data accessible to researchers. The combination of those two factors has set the stage for the FinnGen study, which began in the fall of 2017 and will continue for six years. FinnGen is a consortium being coordinated by the University of Helsinki in which Pfizer and six other pharmaceutical partners will work with Finnish biobanks to conduct genomic and phenotype analysis involving up to 500,000 patients.
Key to this study is that the Finns underwent repeated population bottlenecks (severe and rapid reductions in population size) — including major episodes about 4,000 years ago and again 2,000 years ago. In addition, the Finns were relatively isolated, making the country’s population of 5.5 million today relatively homogeneous genetically. These features make the Finns well suited to gene mapping studies. In such a population, a genetic mutation in one of the population’s “founders” — that is, the small group of ancestors who survived the bottlenecks — can be more easily tracked.
“In both the demographics and the migration history of the population, there are some unique characteristics in how that presents itself in genetics,” says Melissa R. Miller, Senior Manager, Computational Target Validation, Target Sciences and Technologies at Pfizer and one of the Pfizer investigator(s) for the FinnGen study.
A Brief History of the Finnish People
The area that is Finland today was first inhabited by humans after the glaciers from the last ice age receded about 10,000 years ago. But it remained sparsely populated until about 2,000 years ago, and even then, it wasn’t until the 1500s that people began migrating from the early settlements in the southwest of the country — mostly along the coast of the Baltic Sea — into the northern and eastern regions, with those migrant populations often forming isolated enclaves of fewer than 50,000 people. In addition, there have been repeated population bottlenecks, the last one in the early 1700s.
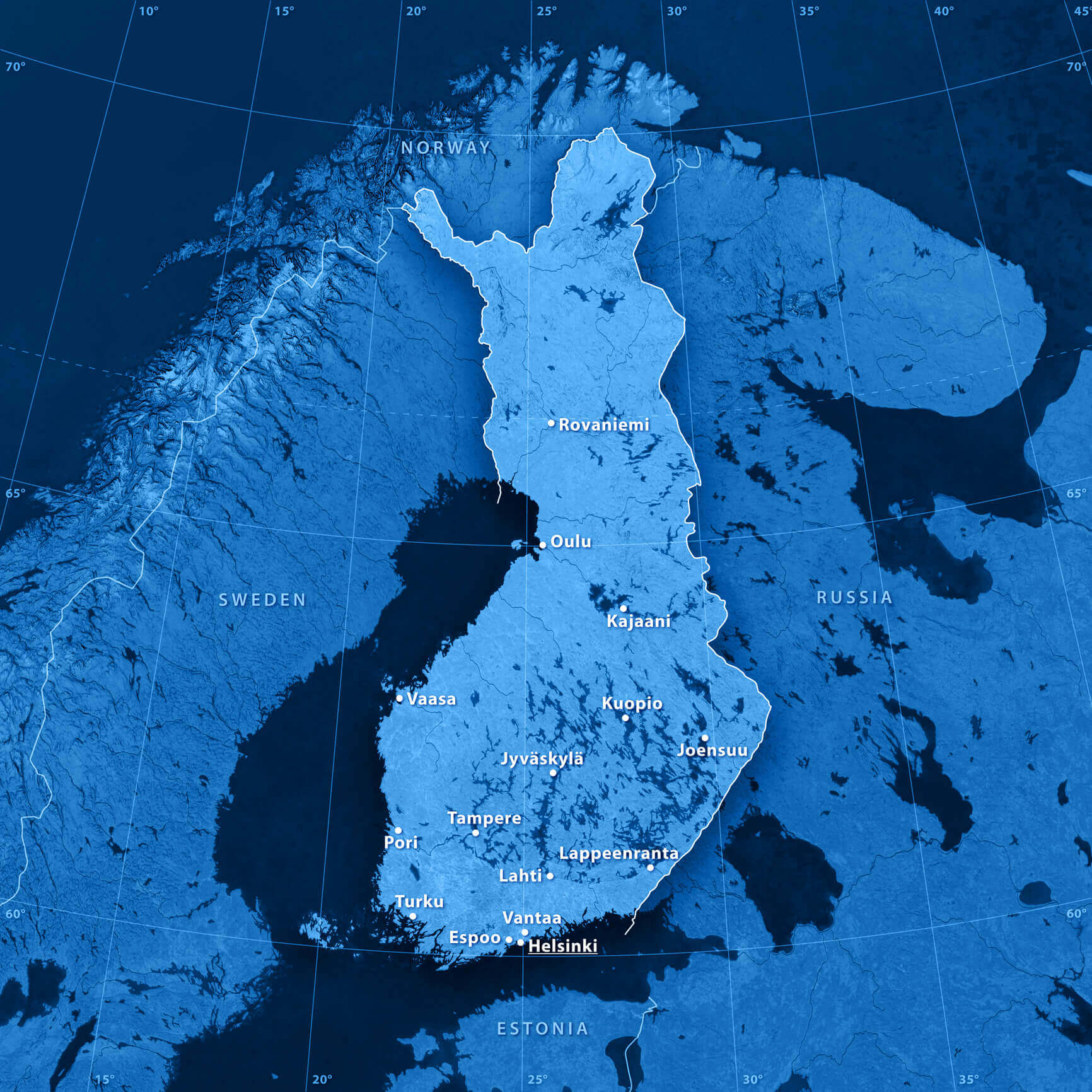
It wasn’t until the 1500s that people began migrating from the early settlements in the southwest of Finland – mostly areas bordering the Baltic Sea – into the northern and eastern regions.
Why the Finns’ Heritage Lends Itself to Genetic Studies
The combination of population bottlenecks and isolation, especially in eastern Finland, set the stage for what has come to be known as the Finnish Disease Heritage (FDH) — more than 35 recessive genetic disorders prevalent in Finland but rare elsewhere.
“Genetic variants that were present when Finland was founded tend to become more frequent over time within the Finnish population compared to the frequency in other non-Finnish populations,” Miller explains.
By studying the DNA of people with those variants, scientists can map the genes linked to a particular disorder with high resolution using what is known as linkage disequilibrium analysis, which provides information about a population’s genetic history that may influence future genetic processes, such as natural selection.
“This will be an opportunity to help broaden the spectrum of human biology that drives drug discovery,” Miller says, noting that it could also inform how clinical trials are designed for new medicines. “We hope to create a more holistic understanding of how genes control different aspects of our health and wellbeing.”
Furthermore, Finland has a unique biobank law that links biological data to other national health, demographic, and social databases, and the law also makes this information available to researchers, creating a powerful tool for further scientific studies.
Linking those databases allows researchers to see, for example, at what age a person starts to take a certain medication or how long after the diagnosis of Alzheimer’s disease a person moves to a long-term care facility. “FinnGen benefits from these large interconnected databases and biobanks,” Miller says. “The hope is that we’ll be able to combine that information in such a way that we will improve our understanding of the trajectory of those diseases and identify factors that contribute to their progression.”
And while the study utilizes the unique genetic heritage of the Finnish population, the results of the study will be relevant to populations all over the world, Miller says.