Checking Our Gut Microorganisms’ Disease-Fighting Might
New gene sequencing tech is helping researchers harness the bacteria, viruses, and fungi that live in our bodies to fight disease.
At this very moment, trillions of bacteria and other germs are swimming in your gut and crawling on your skin. While the thought may make you cringe, these microscopic squatters — collectively known as the human microbiome — are essential to our survival. And now a growing body of research shows how this microbiome could point the way to new treatments and a deeper understanding of disease.
It’s Not All Genetics
For most of the past few decades, scientists have focused on uncovering the genetic underpinnings of disease. What has emerged is the recognition that the human genome doesn’t hold all the answers.
“While extremely important, it’s not all genetics,” said Arpita Maiti, director of External R&D Innovation, Inflammation & Immunology at Pfizer in Kendall Square in Cambridge. “Researchers are very interested in the environmental factors— the things we come in contact with. And one of those things is the trillions of microbes that live within us.”
And as medicine shifts away from over-reliance on antibiotics, scientists are seeing that preserving a healthy balance of internal bacteria is vital to health. “When antibiotics came on board 70 or 80 years ago, they were revolutionary,” Maiti said. “But maybe they were too successful, killing the good along with the bad bugs. Now, we’re dealing with the after effects of antibiotic use and insufficient use of preventative treatment like vaccines, which has made us realize that good bacteria may have a role to play in maintaining health and preventing disease.”
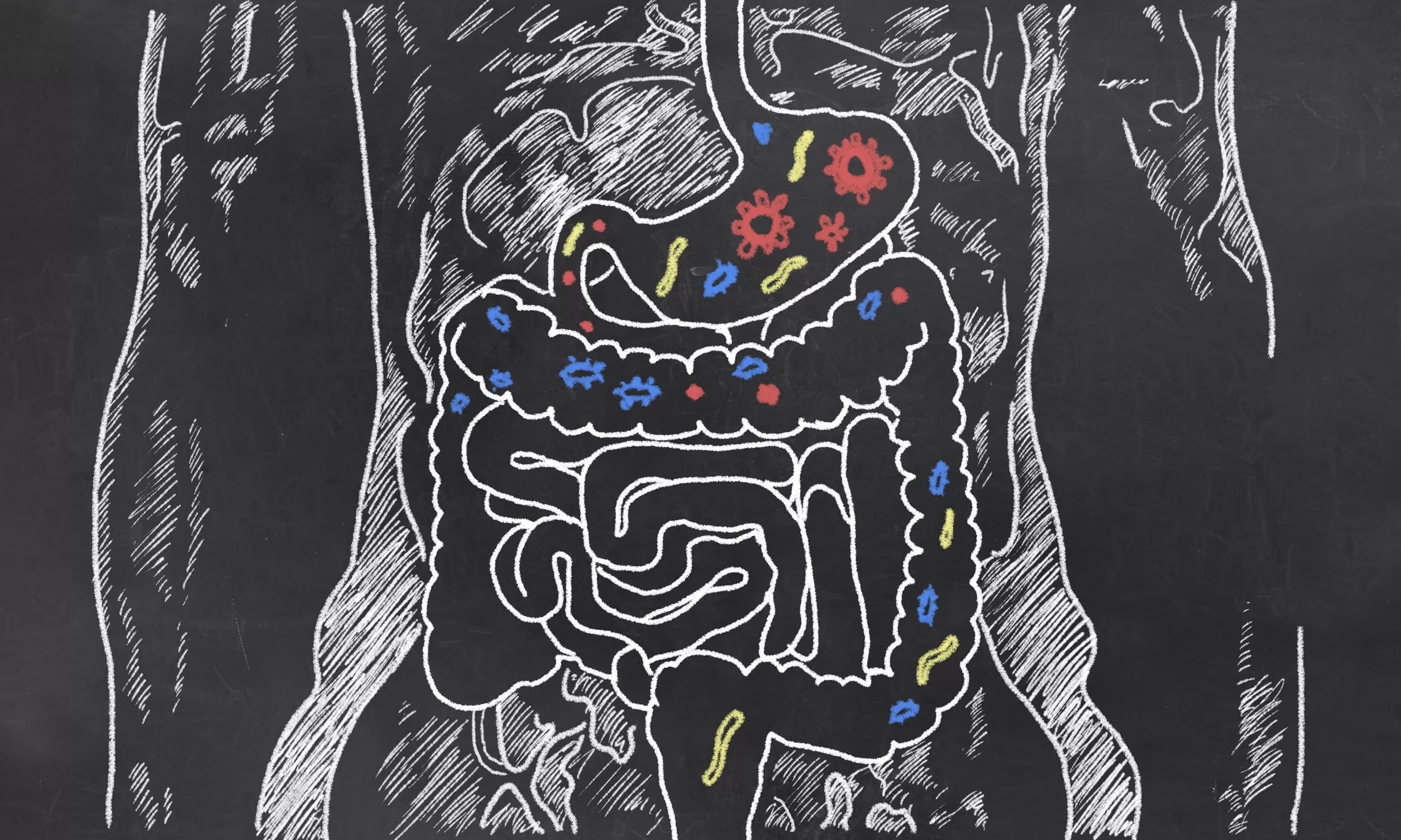
While microbes exist throughout the human body, research has focused on the gut because of the relative ease of collecting samples from that part of the body. In addition to a new wave of biotech startups mining human microbes for innovative therapies, certain companies and initiatives now offer consumers an easy way to have their personal gut bacteria analyzed.
On the therapeutic side, in 2014 Pfizer partnered with the San Francisco startup Second Genome to use its microbiome discovery platform, which uses mixed-species DNA sequencing technology and big data methods to look for disease triggers and possible therapies. Already, collecting microbiome samples is becoming a standard part of most large clinical trials for inflammatory bowel disease (IBD), Maiti said, adding that it could play an even greater role in helping to predict response to treatment and select patients for clinical trials.
Gut Checks and Balances
But researchers are also looking for connections outside the gut. A recent Caltech study found that Parkinson’s disease may be triggered by chemicals released by gut bacteria. The breakthrough findings suggest that an imbalance in these gut chemicals activates an immune response in the brain, leading to neuron damage and deterioration in motor skills. And there’s growing evidence indicating a link between gut bacteria and obesity
and metabolic disease.
“We’re just scratching the surface of understanding these connections,” said Maiti. “But, as we survey more patients and compare the microbiomes of healthy populations to bacteria in disease populations, we’ll be able to take more of a precision-medicine approach in monitoring patients and their response to medicines and in determining how to get the right medicines to the right patients.”