Meet the Team: Homegrown Therapies Get Ready to Fight NASH
One of the biggest public health problems of our time is likely soon to face off with a new wave of cutting-edge medicines. As non-alcoholic steatohepatitis (NASH), a severe form of non-alcoholic fatty liver disease, or NAFLD, continues to be a growing public health threat, scientists are racing to develop treatments. There is currently no FDA-approved medicine for NASH, but nearly 50 candidates, including three from Pfizer, are in clinical testing — one or more which will hopefully be the next breakthrough medicine. And NASH treatments can’t come soon enough: By 2020, NASH is expected to be the leading cause of liver transplants in developed countries.
NASH is a progressive form of NAFLD, developing in about 20% of NAFLD patients when excess of liver fat is accompanied by inflammation, cell damage, and sometimes fibrosis or scarring. Scientists don’t know why some people with milder forms of NAFLD go on to develop NASH, but the condition is closely related to obesity, diabetes, and other metabolic disorders and, in some patients, there appears to be a genetic predisposition.
Homegrown science
In the quest to develop treatments, Pfizer scientists are taking an expansive tack by going after some of the root causes of the condition. While NASH drugs in development at other companies address the inflammation and fibrosis caused by NASH, Pfizer’s approach “is focused on the underlying metabolic drivers for the disease,” says William Esler, a Senior Director in Pfizer’s Internal Medicine Research Unit, based at the company’s Cambridge, MA research site. Esler explains that “by going after what we believe to be the root causes, we hope to see NASH resolve and possibly see a decrease in fibrosis.”
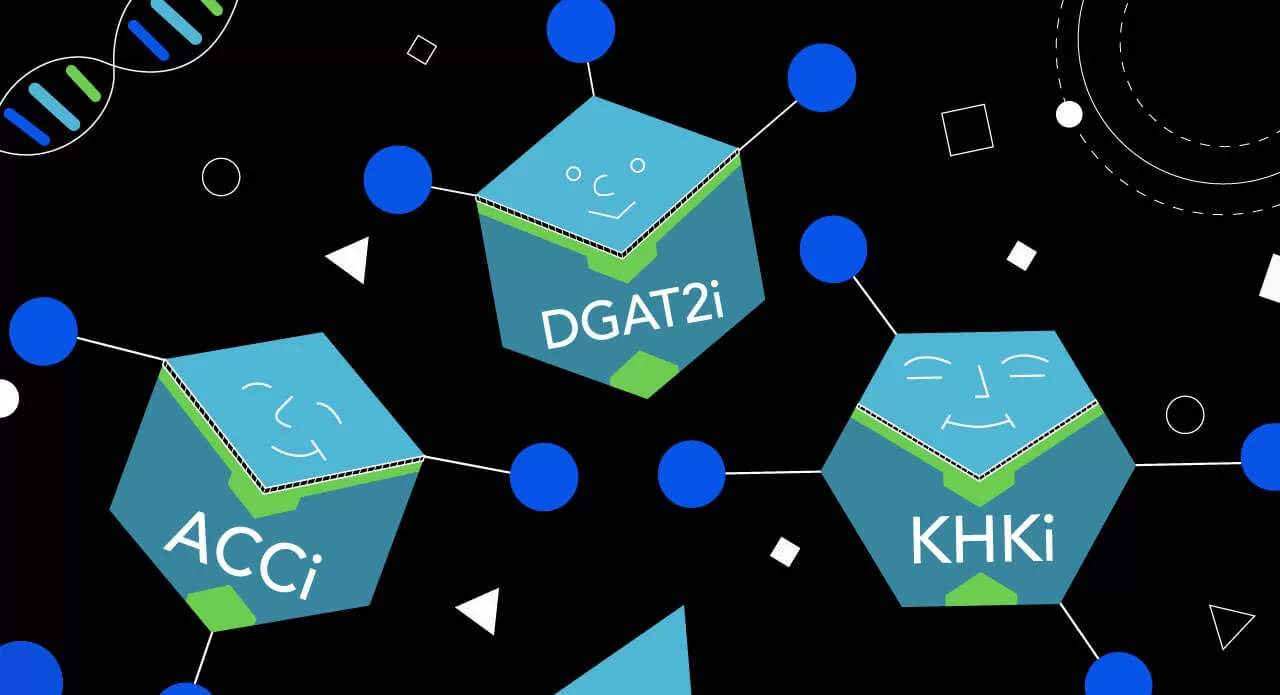
Just as any good sports team needs a deep bench of talented players, Pfizer’s current roster of NASH drug candidates, all of which are based on homegrown science, target different parts of the body’s metabolism. “Each of these drug candidates were derived from science in our own laboratories, and each of these compounds was designed and synthesized by our chemists here at Pfizer,” says Esler.
Read on to learn about more about Pfizer’s NASH “team”:
ACCi

Full name of target enzyme: Acetyl-CoA Carboxylase (“ACC”)
How it works: The first “team member” is an ACC inhibitor. ACC is an enzyme involved in the early steps of fat synthesis. When we eat an excess of carbohydrates, the body’s essential energy source, some of these carbohydrates are converted to fat and may be stored as such throughout the body as well as in the liver. For people with a high-caloric diet, excess fat begins to accumulate in the liver. But by blocking ACC in the liver, fat synthesis can be halted. An ACC inhibitor may also help promote the oxidation — or burning — of existing fats in the liver. “By clearing the liver of fat, our hypothesis is that it will lead to improved or reduced inflammation, says Esler.
Targeting disease drivers: ACC is considered unique because it can potentially impact all three drivers of the disease: lipid accumulation, inflammatory response, and fibrosis.
DGAT2i

Full name of target enzyme: diacylglycerol acyltransferase 2 (“DGAT2”)
How it works: Initially developed as a potential anti-hyperlipidemia medicine, the next “team member” is an inhibitor of DGAT2, an enzyme involved in the final steps of triglyceride synthesis in the liver. When excess carbohydrates and other nutrients are converted to fatty acids in the liver, they are packaged into triglycerides so they can be stored in the liver or secreted. By inhibiting DGAT2, the final step in creating triglycerides is blocked. In addition to this direct action on triglyceride production, inhibition of DGAT2 also indirectly reduces the expression of multiple lipogenic target genes. “This mechanism is extremely encouraging,” says Robert Dullea, a Senior Director in Pfizer’s Internal Medicine Research Unit, also based in Cambridge.
The power of teamwork: Because ACC and DGAT2 work on opposite ends of the metabolic pathway, scientists are testing them in combination to see even greater reduction in liver fat. “We currently have a Phase 2a study ongoing, and we’re very excited about that potential combination,” says Esler.
KHKi

Full name of target enzyme: Ketohexokinase (“KHK”)
How it works: Unique among its teammates, our third “team member” is a KHK inhibitor which can change how the body uses fructose, a ubiquitous sugar used to sweeten foods in the modern diet. Fructose consumption is known to ramp up the synthesis of fat in the liver in an unregulated manner. By blocking the KHK enzyme and stopping the first committed step of fructose metabolism, scientists expect to see a reduction in liver fat and ultimately a benefit in NAFLD and NASH. “Targeting the metabolism of a single, but important, dietary ingredient makes this project unique,“ says Thomas Magee, a Senior Director in Pfizer’s Internal Medicine Research Unit, also based in Cambridge.
Identification of the target: A preclinical model of NAFLD/NASH demonstrated that knocking out the KHK gene resulted in protection from liver fat accumulation and related pathologies such as insulin resistance that can result from excessive fructose consumption.