Phenotypic Drug Discovery Modern Successes
Scientists return to a classic model of drug discovery in an attempt to find novel medicines, while adding a modern spin.
For the past 30 years or so, the main method for finding new medicines has been based on sophisticated knowledge of a specific target — a protein, such as an enzyme or receptor. Once a target for a disease is identified, scientists look for a compound that efficiently binds to and modulates the target, either by screening known candidates or by building a compound from scratch that could be a good fit.
But in recent years, an advent of new tools and data sources with which to understand disease biology has led researchers to have renewed interest in another approach to medicine hunting, phenotypic drug discovery. As its name suggest, scientists begin by searching for a compound that causes an observable physical change in a whole biological system mimicking the disease, such as animals or cells. Using this approach, scientists may not fully know or understand the target or targets on which that compound is acting, perhaps because they’re investigating a disease for which there are few known targets. With phenotypic screening, however, they may have something equally important—visible proof that the compound has modified the symptoms or cured the illness.

“If we start testing compounds in cells that closely represent the disease, rather than focusing on one single target, then odds for success may be improved when the eventual candidate compound is tested in patients,” says Fabien Vincent, an Associate Research Fellow at Pfizer’s Groton, Connecticut site, who is among the leading industry voices on this approach. In the drug discovery process, he explains, phenotypic screening can be used either as an alternative to target-based drug discovery to identify drug candidates, or as a complement to it, by conducting genetic screening in phenotypic assays to narrow in on a specific driver of the disease.
The reason, he explains, has to do with awareness of certain “unknown unknowns” that may influence how promising a target is for eventually treating a disease. “With a target selection approach,” he says, “scientists make certain assumptions about how well a target or pathway is connected to a disease.” And those assumptions are tested at each stage of preclinical and clinical research. Certain assumptions, says Vincent, may fail to be supported by the research — and may eventually lead to that clinical candidate medicine failing at some stage of testing.
The advantage with phenotypic screening is that you’re testing in more realistic systems — disease relevant cells and stimulus — and, thus, it is anticipated that results may be more predictive of the clinical outcome.
The renaissance began with a 2011 review paper showing that phenotypic drug discovery has led to more first-in class medicines (i.e. completely novel drugs) compared to a target-based discovery program. Between 2000 and 2008, of the 50 first-in class small molecule drugs discovered, 28 came from phenotypic strategies compared to 17 coming from the target-based approach, the review found. “This was a big surprise to the industry,” says Vincent. “Because the vast majority of resources had been put in target-based programs.” The drawback of relying exclusively on the target-based approach, adds Vincent, is molecules developed in early discovery don’t often translate into complex models and humans.
Not Your Grandfather’s Drug Discovery
In the early part of the 20th century, chemists developed novel compounds in their labs and would literally walk them over to the pharmacology lab, where they would then be tested in an animal to look for their effects. But today’s approach is “not your grandfather’s drug discovery of the 1960s,” says Vincent. “We’re using very modern tools such as high content imaging, RNA profiling and CRISPR.”
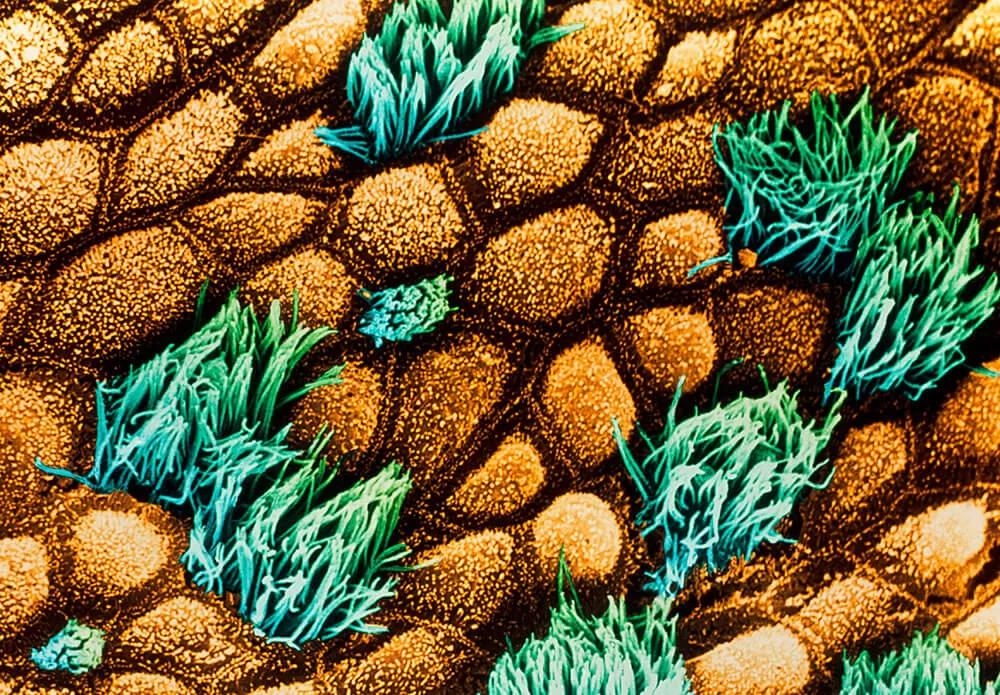
In modern phenotypic drug discovery, the main models are not animals, but cells. The challenge here, however, is producing cell models that are as disease-like as possible. “We really need to make sure that these cell models are of high value and not just some random cell line,” says Vincent. “We need to find a way to recreate the disease in a microplate. That way we can expect higher translation to patients.”
The best predictive models are those built when scientists have access to cells from patients with the disease. “We know they come from someone who actually had the disease, so you know it’s as relevant as possible,” he adds.
A recent example is a phenotypic screen that Pfizer colleagues created for cystic fibrosis (CF), a genetic disorder of the lungs. Patients with CF have a genetic mutation that removes a thin layer of liquid atop lung cells, which is necessary to clear mucus for proper lung function. “Using the right cell type sourced from CF patients, scientists are finding compounds that can re-establish the thin film of liquid,” says Vincent. “That gives you confidence that these compounds will perform the same in patients.”
Ideally, they would hope to someday build models that mimic much more complex diseases like Alzheimer’s or inflammatory bowel disease. “We can’t assume that we know exactly what to do to modulate a disease. But if we start with the disease and we just look for what works, then we have better odds for success,” says Vincent.
Phenotypic approaches may also help discover new biological processes and targets. “In that way it’s very complementary to our other target identification methods,” says Vincent. “You can use phenotypic screening as way to feed the target pipeline.”