Why Mitochondria Is The Organelle Of The Moment
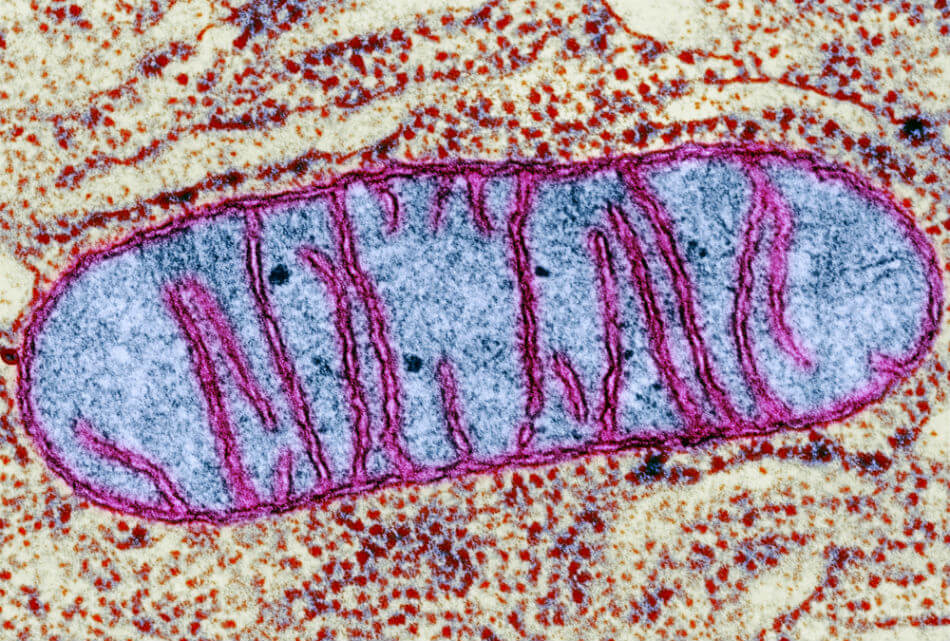
Mitochondria, our cells’ energy converters, have become the focus of many areas of disease research.
As the power plants in virtually every human cell (as well as animal, plant, and fungi cells), mitochondria play an essential role in creating energy to drive cellular function and basically all of our biological processes. Nearly all cells have these sausage-shaped structures, but muscle and nerve cells, which require more energy, have the highest concentrations of mitochondria, numbering in the thousands.
Beyond their role as energy converters, scientists have also recently uncovered how mitochondria play other critical roles: They act as storage tanks for calcium ions, which are involved in blood clotting and muscle contraction; produce the iron compound needed for red blood cells to transport oxygen; and, last but not least, they’re being studied as the lead “executioners” of cells, with the ability to trigger cell death.
Because mitochondria are essential to so many cellular processes, when anything goes wrong with these organelles, they could be linked to many diseases. Recent discoveries have connected malfunctioning mitochondria to Parkinson’s, heart disease, diabetes, and cancer. In addition, since mitochondria have their own DNA, they’re also linked to some rare but devastating genetic diseases that affect the brain, heart, liver, and other key organs.
Ancient DNA
More than a billion years ago, mitochondria were once primitive bacteria cells with their own DNA that were swallowed up by a larger cell. Rather than be digested, they stayed living in the host cells, developing a symbiotic relationship, providing the energy that allowed cells to evolve into complex organisms.
While most of its original DNA has disappeared, human mitochondria today have 37 genes, which are only inherited through the mother. In a recent groundbreaking procedure, a mother who carried faulty mitochondrial genes for Leigh syndrome, a neurological disorder that is fatal, gave birth to a baby using healthy mitochondria from a donor. She previously had two children who died due to Leigh syndrome. Using a controversial technique, doctors transferred the nucleus of the mother's egg cell to the egg of a donor with healthy mitochondria, and then fertilized it with the father's sperm. In effect, the baby has three genetic parents: the mother, father and the woman who donated her mitochondrial DNA.
A Defect in the Mitochondria Recycling Process
Since the 1980’s, scientists have understood that defects in mitochondria may be linked to some forms of Parkinson’s disease, but researchers at Stanford University Medical Center may now have a better understanding of how this happens. As mitochondria covert energy, they go through a lot of wear and tear and produce toxins. Normally the cells take these damaged mitochondria, tag them with certain proteins and then recycle them through a process called autophagy. But scientists have found that a defect in this removal process is leading the damaged mitochondria to emit toxic free radicals that cause nerve cells to die, triggering the symptoms of Parkinson’s.
While these findings are still in early stages, they could eventually lead to early tests for Parkinson’s disease and potential therapies to help repair the process of cleaning out used-up mitochondria.
