Treating Cancer by Using Epigenetics, the ‘Software’ of Our Genes
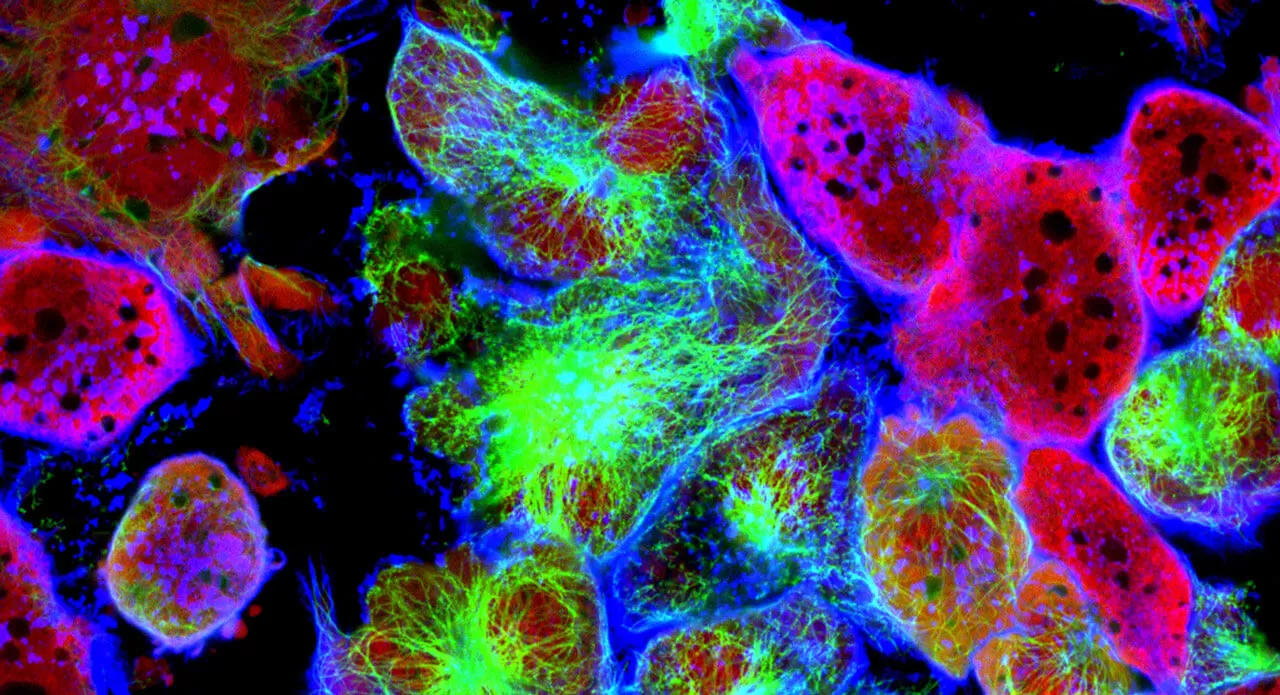
All of our cells possess the same set of DNA. So, why is it that some cells turn into skin cells while other cells turn into lung cells — and still other cells go rogue and turn into cancer cells?
The key lies in the epigenome — the naturally occurring chemical markers that accompany your genes and act as molecular switches that can turn a gene on or off without changing your DNA itself. For example, in agouti mice, even identical twin siblings with the same DNA can have different fur colors when the epigenome is chemically tweaked. These epigenetic markers are established during fetal development but can be also influenced by environmental factors such as nutrition and lifestyle over an individual’s lifetime.

In agouti mice, even identical twin siblings with the same DNA can have different fur colors when the epigenome is tweaked. This golden-yellow mouse on the left is the mouse with the tweaked epigenome. (Courtesy of Randy Jirtle and Dana Dolinoy)
One area of focus for the field of epigenetics (translated literally, “on top of” genetics) is to understand how the epigenome affects human health. As Dr. Shikhar Sharma, a Principal Scientist at Pfizer’s La Jolla, California, site and an expert in epigenetics, puts it, “The best analogy is to think of DNA as the hardware of genes, with the epigenome playing the role of the software on top that directs the genomic hardware, dictating when and where to use certain set of genes or which gene to use, when to use and how much.”
“When the software goes wrong, you can have situations where certain cells can actually be put into a mode where they proliferate, or multiply [uncontrollably], where they don't know when the cell’s life cycle needs to be stopped,” Sharma explains.
What the Epigenome Controls
Epigenetics doesn’t change the DNA itself but it does control the signals that instruct each cell in our body regarding the kind of cell it should become — known as differentiation — and how it should grow or proliferate. Those things have major implications in oncology because if you control the software for cell differentiation and proliferation, you may be able to shut down rogue cancer cells.
Unlike healthy cells that follow clear instructions on what kind of cell they should become and that have a defined lifespan, “a lot of aggressive tumors in general are very poorly differentiated, and they grow uncontrollably,” Sharma says. Because epigenetics plays a key role in giving signals that determine cell differentiation, it has been found to play a major role in driving proliferation of such tumors, he says.
In addition to often growing in an uncontrolled fashion, the problem with poorly differentiated tumor cells is that they are often resistant to traditional chemotherapy and targeted drugs because their shape-shifting nature makes them hard to target with medicines. Manipulating an epigenetic target on cancer cells can make these cells more susceptible to anti-cancer drugs, including chemotherapy.
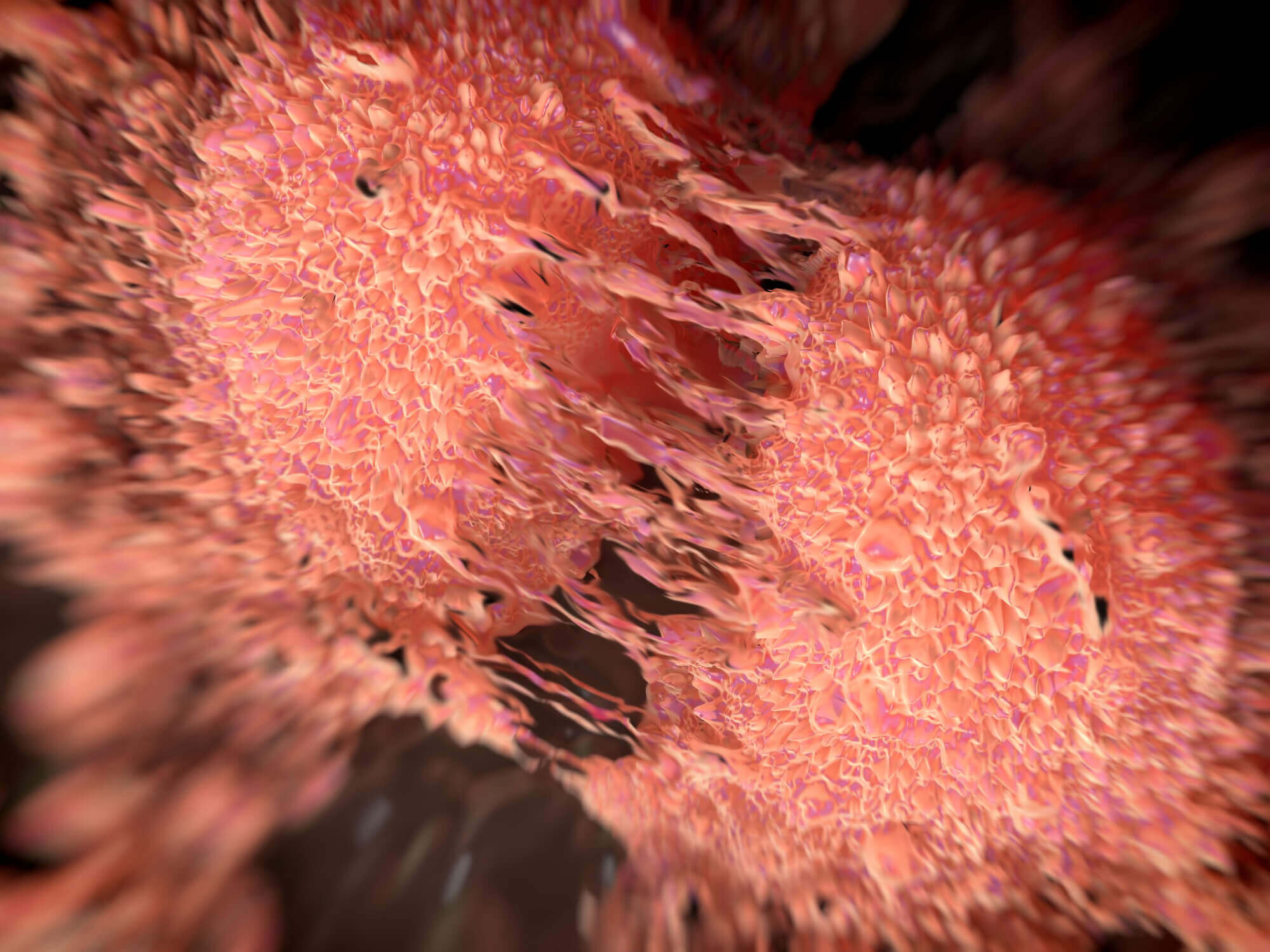
Cancer cell division of two prostate cancer cells in the final stage of cell division (cytokinesis). The cells are joined by several thin cytoplasmic bridges.
How to Manipulate Epigenetics
Traditional chemotherapy works by attacking the DNA of the tumor cells. But because tumor cells share a lot in common with healthy cells, chemotherapy can have unwelcome side effects because it also damages healthy cells. Unlike chemotherapy, epigenetic therapies don’t change the DNA itself, but instead target the signals that turn certain genes on or off, Sharma explains.
A mutation in DNA can cause cancer, but cancer can also originate when epigenetic signals go awry. Importantly, unlike mutations in the DNA, epigenetic aberrations are potentially reversible. The goal of epigenetic therapies is to restore normal gene function by resetting the switches that turn genes on or off.
So, how do you control epigenetics? Some of the most well-studied ways of modifying epigenetics in mammals is to inhibit epigenetic modifiers that turn off genes involved in regulating cell growth and differentiation from expressing themselves—a key process to keep uncontrolled proliferation in check.
For example, one of the methods that Sharma and other researchers have been looking at is a way to control a target called EZH2, which is associated with certain non-Hodgkin’s lymphomas harboring EZH2-activating mutations and several solid tumors that over-express EZH2. When EZH2 is overly active in such lymphoma cells, it interrupts normal cell differentiation, allowing these cells to rapidly proliferate. So, researchers like Sharma are trying to find ways to stop that process.
Once you inhibit EZH2, the associated genes that promote normal cell function and differentiation should start to express themselves again, inhibiting cancer cell proliferation. Inhibition of EZH2 could also make such cancer cells become sensitive to chemotherapy and targeted drugs again, thus restoring their response to conventional cancer therapies, Sharma explains.
The Challenges and Advantages of Epigenetic Therapies
The foremost challenge facing epigenetic therapies is how to identify exactly which patients will benefit from a compound and which will not. The central question, Sharma says, is “Can you find specific gene patterns that can predict sensitivity to a compound?”
In the case of EZH2, for example, there isn’t a test (other than for non-Hodgkin’s lymphoma) that can identify a marker that would tell doctors that a certain patient would be a good candidate for epigenetic therapy.

A key question in epigenetic therapy research, says Shikhar Sharma, "is whether you can find specific gene patterns that predict which patients will respond and which will not."
“So, most of the researchers are trying to figure out if over-expression of EZH2 by itself, or a signature of downstream genes it controls, can be predictive,” Sharma says, noting that further understanding of the role of epigenetic modulators in the immune response as well as their function in tumor growth would also be critical. But it's not easy to find a convenient marker that can determine which patients might respond well when treated with EZH2 inhibitors, he adds.
For most cancer patients, epigenetic therapy will most likely be used in combination with chemotherapy and/or targeted drugs. For example, in certain solid tumors, EZH2 was recently shown to drive resistance to standard-of care (SOC) chemotherapy and targeted therapy agents, so using epigenetic therapy in combination with such SOC agents could improve the response, Sharma says.
“A lot of tumors in general become resistant to chemotherapy with time,” he notes. “And a lot of patients don't tolerate chemotherapy very well.”
Unlike chemotherapy, which attacks the DNA itself in both tumors and in healthy cells, the medicines that affect epigenetics aim to restore normal gene expression patterns in cancer cells. “Comparatively, it could be less toxic,” Sharma notes. “So, it has the potential to have fewer or less severe side effects in patients.”





